Platform Capability Start With Quality.
Lock with Confidence.
Unified study oversight and analytics powered by harmonized data, intelligent dashboards, and real-time insights — all in one validated platform.
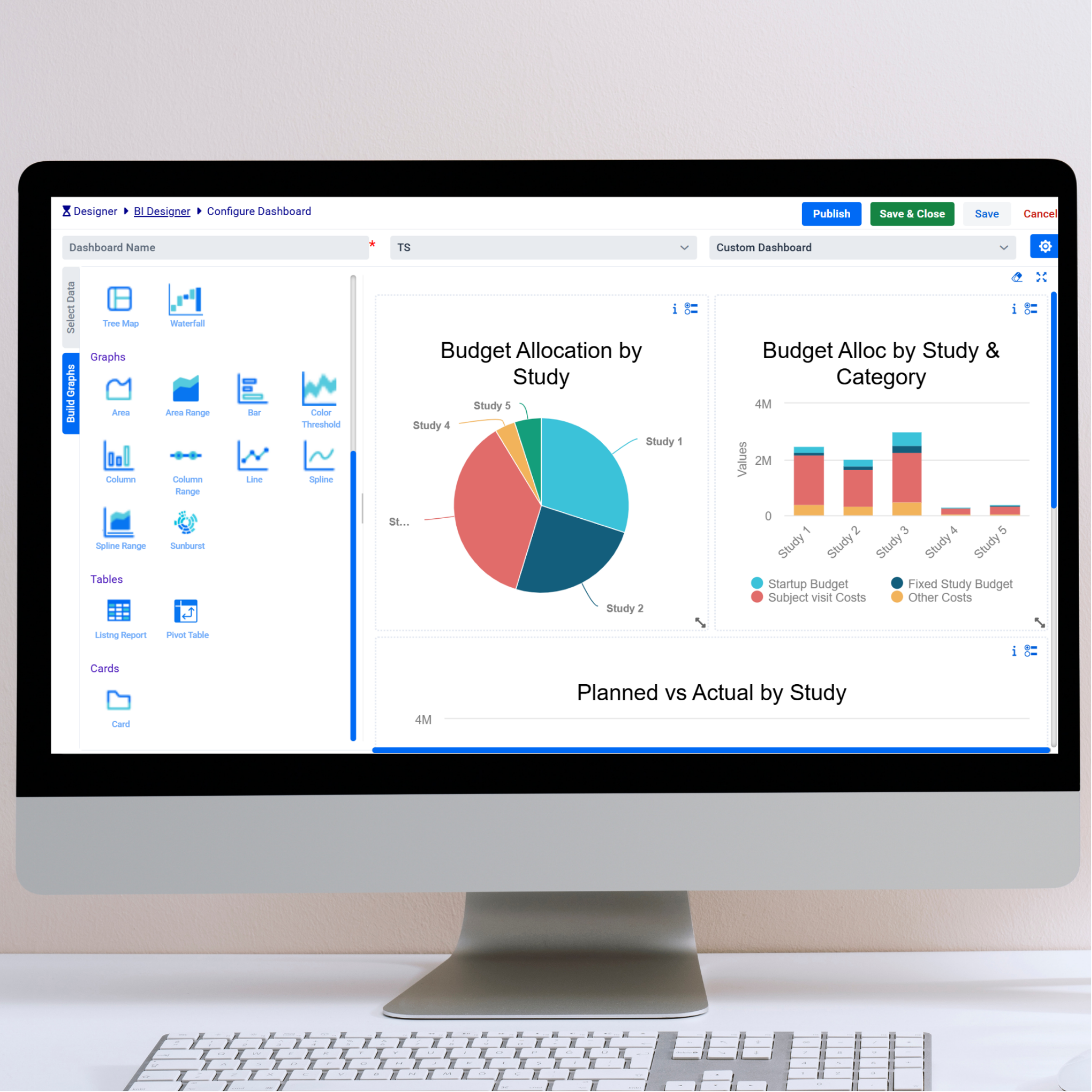
Modern Data Management for Modern Trials
ClinDM™ transforms clinical data management by embedding Agentic AI and automation across the data management lifecycle — from protocol-driven planning to real-time discrepancy management — so data managers can focus on high-value decision-making instead of manual reviews and tracking.
Protocol-driven planning with Agentic AI. Extract CtQs and Data checks directly from the protocol and bring ICH E8 (R1) principles to life by building quality directly into DM planning workflows.
Always-on data surveillance. Deploy ML models that continuously monitor aggregate data to surface anomalies, trends, and new areas of risk.
Faster review cycles, cleaner outcomes. Reduce manual review effort by >50% while improving consistency, traceability, and quality through auto-query functionality and digitized clean patient tracking.
DVP Agent Screenshot
Let Agents Automate Study Build
With Agentic AI, Data Managers can:
-
Cut database build from 12 weeks to 2 weeks with agents drafting your CRF Specs
-
In less than 2 hours create your Data Check Plan directly from the protocol
-
Auto-code complex cross-source data checks and reduce programming time by 60%+
-
Create synthetic test data to validate programmed checks
ML driven data review with automated workflows
During study conduct, automated functionality allows Data Managers to:
-
Identify duplicate patients, digit preferences, lab outliers, and unexpected correlations using pre-built, configurable ML models
-
Automate complex cross-source validation and vendor reconciliation with built-in workflows
-
Write queries directly back to the EDC without leaving the platform
Lab Outlier Screenshot
Real-Time Discrepancy Management and Clean Patient Tracking
ClinDM™ provides data managers a single workspace to review, resolve, and track data quality without jumping between tools.
Unified Review Workspace
Review discrepancies, send queries, and assign follow-up actions in one place.
AI Chatbot Queries
Perform adhoc data queries with Kini™, the platform’s built-in AI chatbot.
Change Tracking
Track new and changed data between review cycles to reduce redundant review.
Workflow Monitoring
Monitor workflows by domain, user, site, and study.
Clean Patient Tracker
Support interim analyses, data snapshots, and database lock.
Scheduled Deliveries
Schedule outbound data deliveries to ensure submission-ready datasets are delivered on time.
Built for Regulated Environments
ClinDM™ is designed for regulated clinical environments where transparency, traceability, and control matter.
Quality by Design
ICH E8 / E6 aligned quality-by-design approach
Traceability
Full traceability of CtQs across data management review activities
Compliance
Controlled workflows with auditability and role-based access
Reuse
Consistent, reusable data checks and analytics across studies and programs
Provide Quality, Reliable Data Without All the Hassle.
See how ClinDM™ helps teams automate the work that slows them down — and deliver cleaner, more reliable data with confidence.